PI kids are asking: Why is rubber stretchy?
There are two connected reasons why rubber is stretchy. One has to do with what rubber looks like deep down inside. The other is a deep secret of the universe.

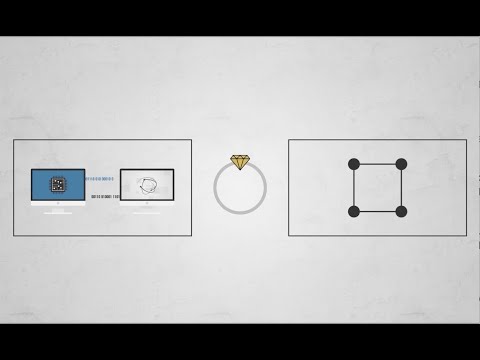
Rubber is a special material because it’s made out of really long molecules. You can think of these molecules like strings of beads. Usually, all of the bead strings lie jumbled up together, like necklaces in a box. But if you pull on them, they can stretch out and get longer, the way necklaces that fit in a little box can end up stretching all the way across a table.
If you pull on a rubber band, the molecules inside will go from looking like this…

… to looking like this:

But then, if you let go of the rubber band, it snaps back. Why? Why doesn’t it just stay all stretched out and nicely arranged?
Here’s the deep secret of the universe part: nature likes things messy. See how the stretched beads are all tidy and lined up, and the unstretched beads are all scrambled and on top of each other? A scientist might call them “ordered” and “disordered.” Nature likes them disordered. Left to its own devices, nature will always make a mess. And when a rubber band snaps back, it’s making a molecular mess.
This tendency of nature to jumble things up is called “entropy.” Entropy is why tidy things jumble up, sliding things slow down, and hot things cool off. And it’s why rubber bands snap back.